Thermodynamics is a very important subject in physical science, which focuses mainly on the mathematical analysis of energy relationships (heat, work, temperature, and equilibrium).
Macroscopically, statistical mechanics explains the various laws of thermodynamics. Thermodynamics applies to a wide variety of topics in biochemistry, science, and engineering, especially physical chemistry, chemical engineering, and mechanical engineering.
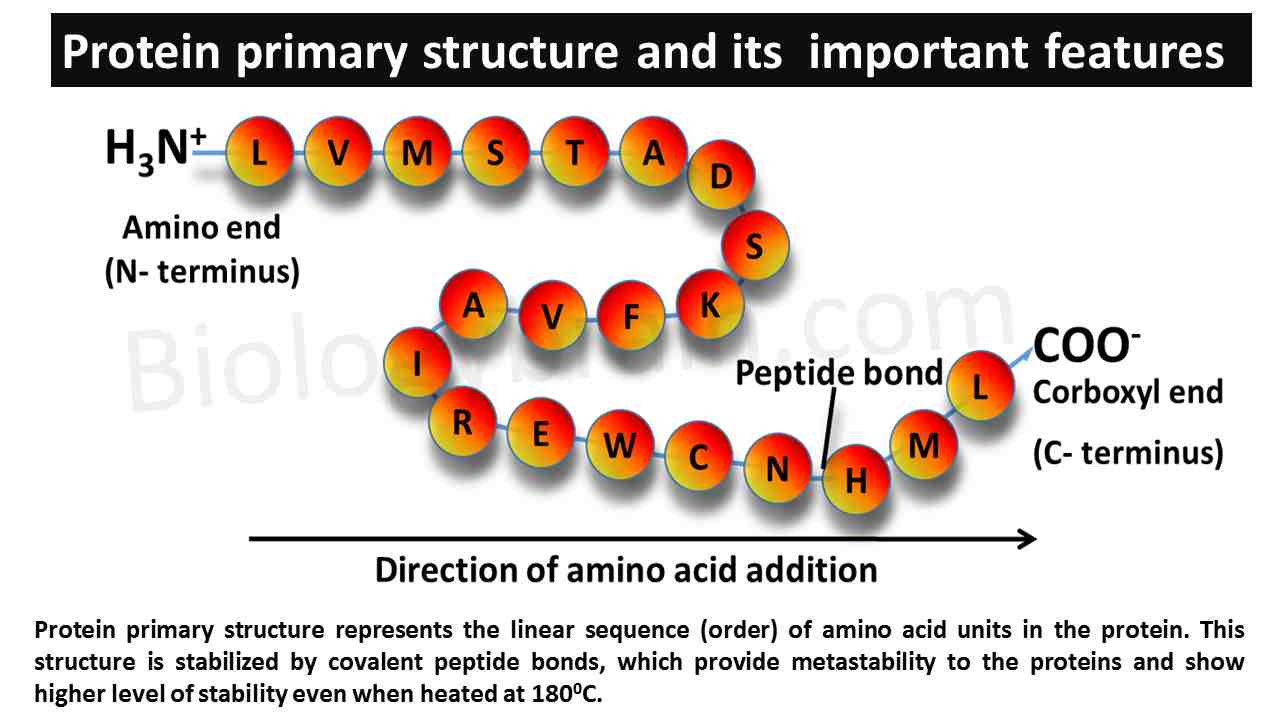
Thermodynamics of protein folding
According to the hypothesis of thermodynamics in biochemistry, the random coiled form of the proteins is the most favorable conformation for retaining biological activities, because it permits the highest degree of disorderliness. If any protein needs to get a particular structural level confirmation, it should first compensate for the enthalpy of the system. The explanation for this concept has been clearly described below.
∆G= ∆H-T∆S
∆G for a process depends on ∆H, ∆S, and temperature [T]
∆G= Free energy change
∆H= enthalpy change
∆S= entropy change
Considering the above formula. If enthalpy (ΔH) is positive, the reaction is endothermic, which means heat is absorbed by the reaction because the products of the reaction have more enthalpy than the reactants. In another case, if ΔH is negative, the reaction is exothermic, which means the overall enthalpy decrease is achieved by the generation of heat.
For example: If we notice an enzymatic reaction, the process can be of two types.
Endothermic reaction:
If any enzyme requires energy to perform the reaction, it means the final product of the reaction will have more enthalpy than the substrate; hence, for the reaction to happen, we should provide the energy. In this process, energy or heat will be absorbed.
Exothermic reaction:
If any enzyme performs the reaction without additional energy, which means the final product of the reaction is having less enthalpy than reactants. In this process, heat or energy will be released into the surroundings.
Hence, for any reaction to perform, the value of the free energy should be negative (-∆G). The proteins in a random coiled form have more entropy (∆S) than any other structure of the protein. This means, higher entropy (randomness or disorder) of the protein provides always a favorable condition unless a greater negative enthalpy value can compensate for the loss in entropy.
The increase in enthalpy of protein structure can be obtained from various interactions such as hydrogen bonding at the intramolecular level, van der Waals forces, hydrophobic interaction, and dipolar interactions (Salt bridges). According to sheer absolute values of enthalpy, the significant increase of enthalpy in the protein structure is obtained from hydrogen bonding than other interactions during protein folding.
If we see the process of protein folding, the initial interaction between two peptide bonds reduces the randomness of the polypeptide strand which is mainly by the formation of a hydrogen bond.
As above mentioned, in a randomly coiled polypeptide strand, the level of entropy is more and enthalpy is less compared to a higher-ordered structure (condensed). Also, the solvent present around the protein forms hydrogen bonds with the moieties in the protein backbone. Once the folding is initiated, the intermolecular hydrogen bonds will be formed by the gain in structure and it should compensate for both the loss in entropy and the loss of hydrogen bonds with the solvent.
During alpha-helix formation, initially, the linear polypeptide chain forms a single hydrogen bond between carboxyl oxygen of i th amino acid and imino hydrogen of i+ 4th amino acid to form one turn, it leads to initial changes in orientation of 6 torsion angles (α, β and ω) in the polypeptide chain.
Formation of additional turn to this initial nucleation site requires only orientation of three torsion angles by hydrogen bonding. Thus, it is more favorable to add turns of a helix to an initial nucleation site and this is described as a cooperative effect in helix formation.
Due to the cooperative effect, the formation of further turns is more likely to follow the order established in the previous steps. The formation of helical structure follows exactly this. The periodicity of fold-formation can arise from sequential interactions in α- helix or long-distance sheet formation in β- pleated sheets.
Frequently asked questions
Which of the following forces is favorable for protein folding?
a) Hydrogen bonding
b) Hydrophobic interactions
c) Vander Waals interactions
d) Salt bridges formation
Answer: B
Explanation: Even though there are so many noncovalent interactions in the protein structure, only hydrophobic noncovalent interactions provide the driving force for the protein folding and keep protein stable and biologically activate.
What are the driving forces of protein folding thermodynamics?
Answer: Mostly, the hydrophobic noncovalent interactions provide the driving force for protein folding, which is thermodynamically favorable.