Thermogenin Definition
Thermogenin is an uncoupling protein found in the plasma membrane of prokaryotes and the inner membrane of mitochondria of both plants and animal cells.
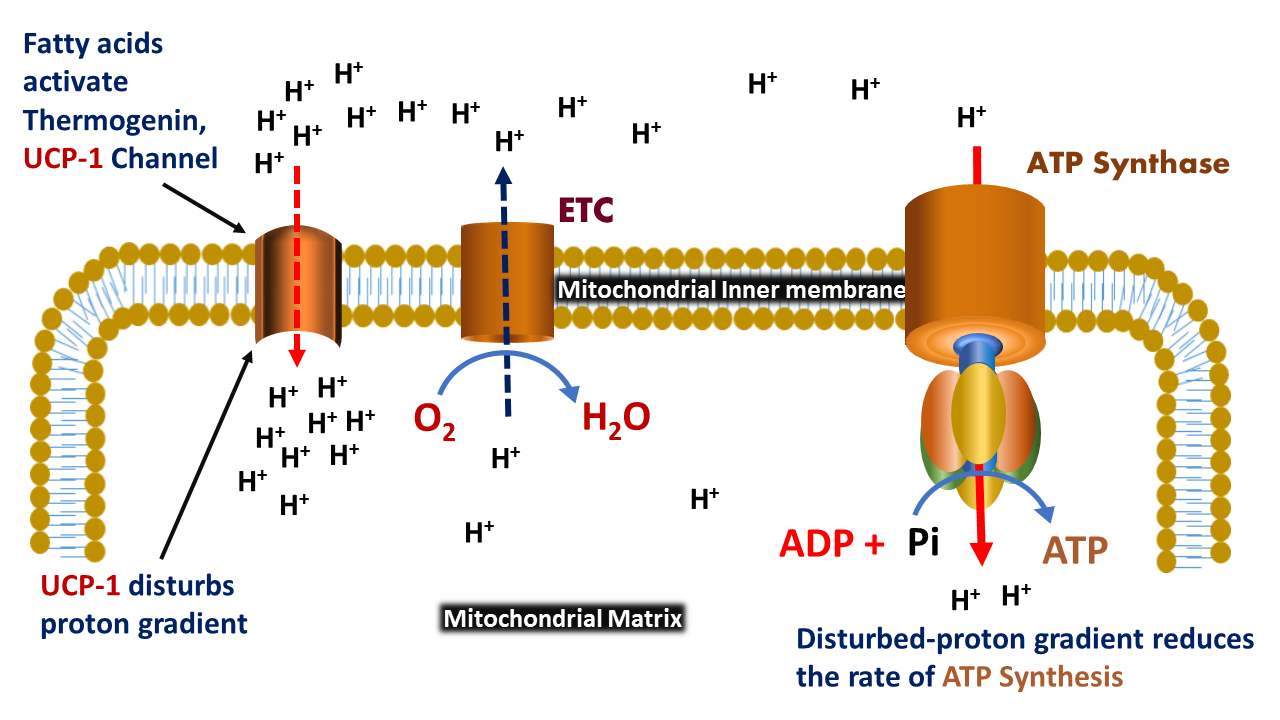
Uncoupling protein is involved in the transport of ions and heat generation (thermogenesis). Thermogenin protein can disturb the proton gradient that was generated by NADH-powered pumping. The three well-known uncoupling proteins including UCP1, UCP2, and UCP3.
Introduction to Thermogenin
Uncoupling protein-1 (UCP1) is found on the mitochondrial membrane of brown adipose tissue (BAT) and that contributes to generating heat by non-shivering thermogenesis, hence the protein termed thermogenin. This uncoupler plays a positive role in newborn babies with generating body heat as they have a high surface area-volume ratio.
Uncoupling protein is a transmembrane protein of the inner mitochondrial membrane that disturbs the proton gradient by increasing the permeability of protons through the inner mitochondrial membrane from intermembrane space to the mitochondrial matrix during oxidative phosphorylation.
The mechanism of uncoupling protein is closely related to other mitochondrial inner membrane proton channels, adenine nucleotide translocator (mitochondrial metabolite transporters) that allows the transport of protons into the mitochondrial matrix from mitochondrial intermembrane space.
Key points of Thermogenin
- Uncoupling protein can be activated by fatty acids and inhibited by purine nucleotides (ADP and GDP).
- The function of uncoupling protein (thermogenin) is similar to the function of adenine nucleotide translocator (inner membrane proton channels).
- UCP1 was found only in brown adipose tissue.
- UCP1 is the only protein that can able to generate heat in a non-shivering way by considering body metabolic conditions.
- The sympathetic neurotransmitter, norepinephrine stimulates UCP1 activity through a signaling cascade.
How does thermogenin generate heat?
Fatty acids in the brown adipose tissue activate the UCP1, while nucleotides inhibit the function of the transporter.
The activation process by fatty acids occurs in the following signaling cascade.
Terminals of neurons in the sympathetic nervous system release the neurotransmitter, Norepinephrine (catecholamine hormone), which in turn actives the Beta-3 adrenergic receptor located on the plasma membrane.
The triggered receptor activates the adenylyl cyclase. This enzyme, in turn, catalyzes the synthesis of the second messenger, cyclic AMP (cAMP) from ATP molecules. Then the produced cAMP activates protein kinase A by releasing catalytic subunits (C subunits) from its regulatory R subunits.
The released C subunits of protein kinase A bind and activates the enzyme, triacylglycerol lipase by phosphorylation. Thus, the activated lipase cleaves the bulk molecules, triacylglycerols into free fatty acids; then, the released fatty acids activate the protein thermogenin, especially, an uncoupling protein-1.
During the termination of thermogenesis, the function of uncoupling protein can be inhibited by purine nucleotides (GDP and ADP), thus the thermogenin is inactivated, and the remaining fatty acids are disposed of through oxidation, which facilitates the cell to return its normal energy-conserving state.
What is the function of Thermogenin?
- Thermogenin involves the sending of protons-return back from the mitochondrial intermembrane space to the mitochondrial matrix.
- Reduces the rate of ATP synthesis by disturbing the proton motive force, which is a need for oxidative phosphorylation.
- Involved in the generation of heat in the brown adipose tissue of newborn babies.
- Promotes the rate of fatty acid oxidation.
- Involved in the regulation of free radicals and reactive oxygen species (ROS).
- Increases the oxidation of NADH molecules to generate an electrochemical gradient across the mitochondrial membrane.
Scientific studies on uncoupling protein
Gene transfer therapy is a current scientific strategy to deliver the specific gene into the target place of the DNA, that encodes uncoupling protein. Moreover, the experiments to enhance the expression of thermogenin in a significant amount to increase metabolic rate in obese persons offers effective treatment options for patients with obesity problems.
Which uncoupling protein is a major contributor to generate heat in brown adipose tissue?
In the journal of Biochim Biophys Acta, Nedergaard, et al., 2001 stated that the expression analysis of uncoupling proteins revealed that UCP1 has uniqueness for generating the heat in brown adipose tissue compared to other uncoupling proteins UCP2 and UCP3. In their studies, they also found the higher expression of the UCP1 gene during cold acclimation (in adults or perinatally) and overfeeding, and reduced expression during fasting in the patients with genetic obesity.
Finally, their study concluded that UCP1 is a unique uncoupling protein from others (UCP2 and UCP3) and is the only protein that can mediate adaptive non-shivering thermogenesis and ensuing metabolic inefficiency.
Data source:
Palou A, Picó C, Bonet ML, Oliver P.The uncoupling protein, thermogenin. Int J Biochem Cell Biol.
Mortola JP, Naso L.Brown adipose tissue and its uncoupling protein in chronically hypoxic rats.Clin Sci (Lond).
Nedergaard J, Golozoubova V, Matthias A, Asadi A, Jacobsson A, Cannon B. UCP1: the only protein able to mediate adaptive non-shivering thermogenesis and metabolic inefficiency.Biochim Biophys Acta.
Golozoubova V, Hohtola E, Matthias A, Jacobsson A, Cannon B, Nedergaard J. Only UCP1 can mediate adaptive nonshivering thermogenesis in the cold. Only UCP1 can mediate adaptive nonshivering thermogenesis in the cold.
Nedergaard J, Golozoubova V, Matthias A, Shabalina I, Ohba K, Ohlson K, Jacobsson A, Cannon B. Life without UCP1: mitochondrial, cellular and organismal characteristics of the UCP1-ablated mice. Biochem Soc Trans.
Matthias A, Ohlson KB, Fredriksson JM, Jacobsson A, Nedergaard J, Cannon B. Thermogenic responses in brown fat cells are fully UCP1-dependent. UCP2 or UCP3 do not substitute for UCP1 in adrenergically or fatty acid-induced thermogenesis. J Biol Chem.