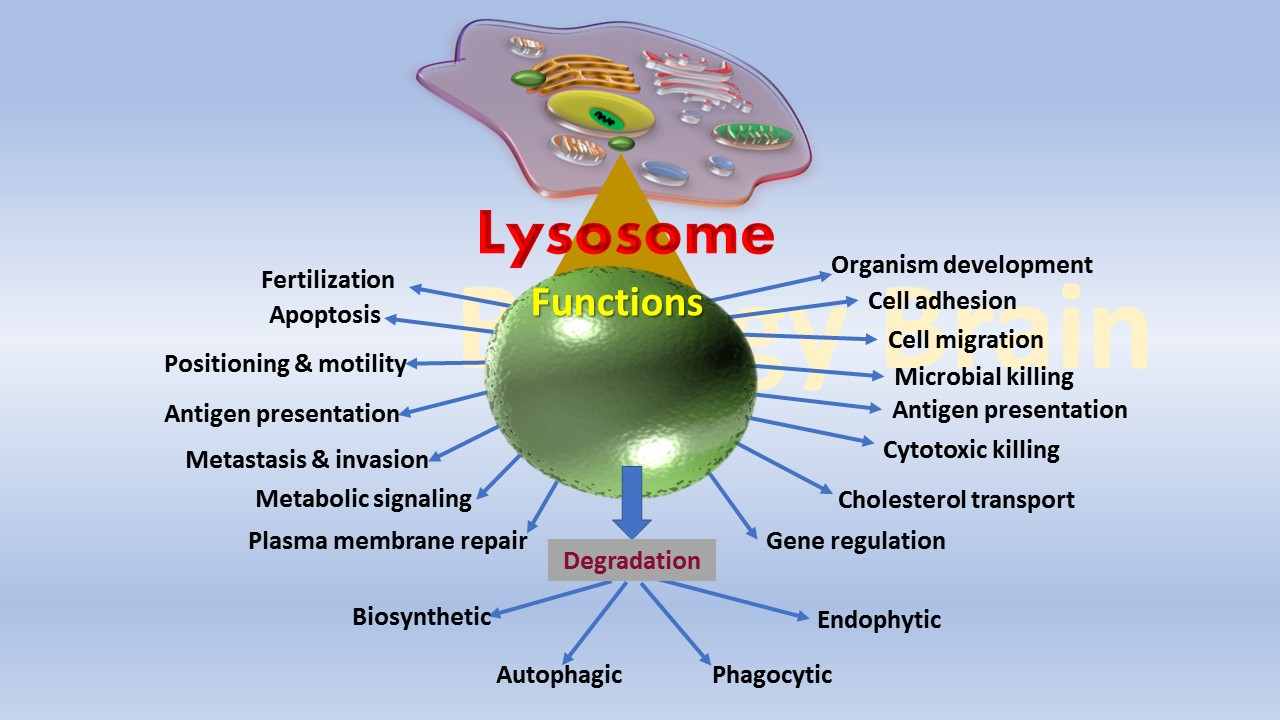
Lysosome Definition
Lysosome is a membrane-enclosed cellular organelle that possesses distinct enzymes involved in the digestion of unwanted or damaged cellular polymers including proteins, DNA, RNA, carbohydrates, and lipids. Lysosomes also play an integral role in the breakdown of pathogenic molecules especially viral particles and organic toxins to reduce the cause of disease. Another function of lysosomes is digestion of the fragments of unwanted or aged cellular organelle such as mitochondrion.
What does the Lysosome do in a animal cell? (Function of Lysosomes)
- The lysosome in the animal cell plays a pivotal role in maintaining cellular homeostasis.
- Lysosomes can digest both intracellular and extracellular components.
- They play a major role in autophagy.
- Lysosomes are involved in crinophagy (secretion), in which the removal of excess secretory granules takes place.
- Lysosomes participate in the dissolution of blood clots.
- The acrosome is a modified lysosome that is located at the tip of the sperm and is involved in fertilization.
- The lysosome is also involved in phagocytosis.
- The lysosome plays a crucial role during starvation in providing nutrients to the body.
- They are involved in the secretion of thyroid hormones.
- They are involved in the development of organisms, e.g. the degradation of tadpole tail during the metamorphous by lysosomal enzymes is a crucial process.
- They are involved in the regression of structures or organs after the periods, e.g. Uterus, Mullerian and Wolffian bags.
- Lysosomes are crucial for the downregulation of membrane receptors in neurons so that they respond less strongly to extracellular ligands.
- Lysosomes are also critical for regulating cholesterol homeostasis and dissolving G-protein aggregates associated with neuropathies like Huntington’s disease (HD).
- The lysosome is an important organelle in the antigen-presenting cells and proteolytic cells. For example, macrophages require lysosomes (scavenging cells of the immune system) to digest pathogenic molecules. Hence, the abnormal macrophages in the immune system are considered a diagnostic marker for Gaucher’s disease.
What are the characteristics of lysosomes?
- Lysosomes are popular as suicidal bags of the cell, and they are referred to as recycle bins of the cell as well.
- The lysosomes are single membrane acidic compartments, that contain a bunch of acid hydrolases.
- Lysosomes are present in animal cells but absent in plant cells, where lysosome function is undertaken by vacuoles or peroxisomes.
- The lysosome contains a highly acidic pH environment compared to the cytoplasm of the cell. This acidic nature of the lysosomes is due to the presence of V-class proton pumps on the lysosomal membrane. They pump the protons from the cytoplasm to the inside of the lysosomes. The process requires energy.
- They have several important roles in cellular metabolism, e.g. they degrade extracellular proteins which are engulfed by endocytosis and the aged materials of cytoplasm such as organelles and cytosolic proteins.
- The lysosome contains more than 50 types of hydrolytic enzymes.
- Lysosomal enzymes are, however, inactive at physiological pH (7.0 to 7.8) and they can catalyze the enzymatic reaction only at their optimal acidic pH.
- If the lysosomal membrane breaks down, the enzymes become inactive at cytosolic pH.
- Acid phosphatase is a marker enzyme of lysosomes because lysosomes can be recognized by staining this enzyme.
- In some cases, cathepsin is also considered a marker enzyme of lysosomes.
- Lysosomes contain an enzyme called labilase that makes the lysosomal membrane labile.
- The lysosomes also contain the molecule cortisone (a stabilizer), which stabilizes the lysosomal membrane.
- Intracellular compartments, such as aged organelles, are taken into the lysosomes by a process called autophagy. For instance, when a mitochondrion comes to the end of its ten-day life, then it is engulfed by a membrane that is derived from the endoplasmic reticulum. The ER membrane-enclosed mitochondrion vesicle then fuses with a lysosome, resulting in the digestion of old mitochondria.
- Several defects in the gene that encodes specific lysosomal enzymes have been discovered. A certain defect in a functional gene may result in reduced enzymatic production, affected enzymatic activity within the lysosomal environment, inappropriate localization of an enzyme within the lysosome, or faulty post-translational modification of an enzyme.
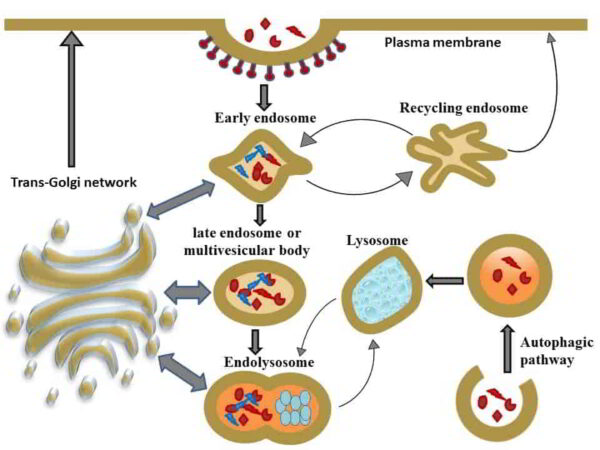
- Newly synthesized lysosomal proteins and enzymes will be delivered to lysosomes from the trans-Golgi network via early endosomes, recycling endosomes, and late endosomes/multivesicular bodies. Following lysosomal fusion with late endosomes, endolysosomes, and lysosomes, these are re-formed by a process called the maturation process.
Degradation of cell membrane or extracellular components by lysosomes
- About 40% of the cell proteome is made up of extracellular proteins, which belong to important protein classes like growth factors, cytokines, and other extracellular virulent proteins that play crucial roles in the development of diseases.
- Therefore, careful monitoring of the activity of unwanted or outdated extracellular proteins and their removal is crucial for cell survival.
- Delivery of these foreign or cellular proteins to lysosomes is very common in the cells.
- Lysosomes are terminal compartments of the endocytic and autophagic pathways.
- Organelles called early endosomes take in solutes and macromolecules from the extracellular space. Sorting this cargo into the cell’s recycling and degradative compartments is the primary function of early endosomes.
- The late endosome is an important location for sorting membrane vesicles and their contents into downstream processes.
- The trans-Golgi and early endosome compartments deliver proteins to the late endosome, and the late endosome produces vesicles that travel to the trans-Golgi or lysosome.
- Whether the protein in the late endosome will be delivered to the lysosome or to the trans-Golgi depends on signal motifs and posttranslational changes such as protein glycosylation.
- If the proteins that need to be broken down are delivered to the lysosomes, then the enzymes, especially hydrolases in the lysosomes, break them down.
- The process of endocytosis typically involves the uptake of protein receptors and their ligands into clathrin-coated vesicles from the surface of the plasma membrane. Then, they are directed to different intracellular compartments after sorting into the early endosome.
- These receptor proteins could either be moved to the recycling endosome, where they would then be exposed once more, or they could be moved to multivesicular bodies, where they would then be moved to late endosomal compartments and lysosomes, directing them for degradation.
Degradation of intracellular components by lysosomes
- Lysosomes engulf and break down cytosolic substrates as a part of the macroautophagy alternative degradation pathway in cells.
- Typically, unwanted cellular proteins are eliminated by the ubiquitin-proteasome system (UPS), but the process of macroautophagy eliminates proteins as well as larger cellular entities like damaged organelles and invasive pathogens.
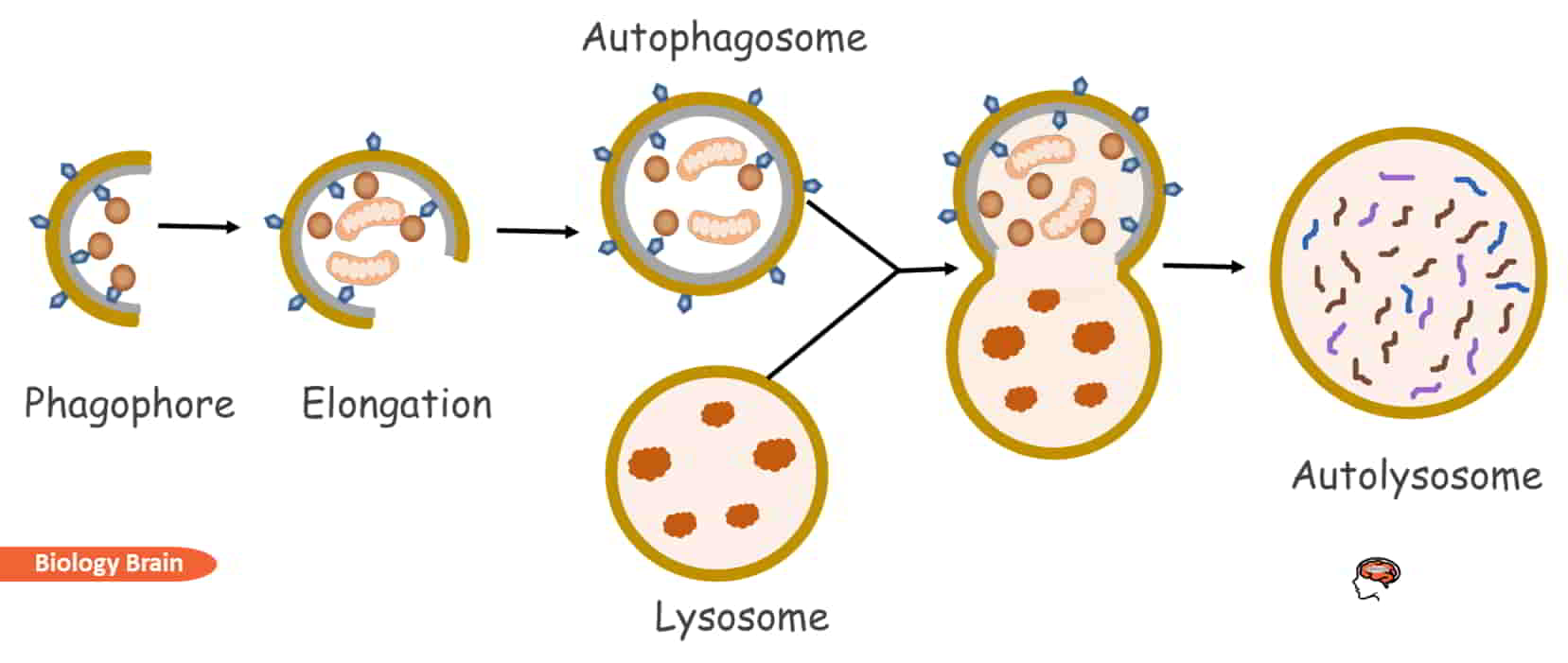
- The macroautophagy process begins with the formation of a phagophore, a separated double membrane, by a complex of different cellular proteins.
- When the cargo is ready to be degraded, the phagophore elongates and sequesters it from the cytoplasm by forming an autophagosome.
- An autolysosome is created by the fusion of an autophagosome with a lysosome after the membrane has been enclosed and matured.
- Then the autolysosome’s acidic conditions and its hydrolase enzymes digest the cargo.
Important cellular components that lysosomes degrade include
- G-protein clusters related to neuropathies like Huntington’s disease are degraded by lysosomal enzymes.
- Multivesicular bodies, which are packed with receptor proteins on the plasma membrane for degradation, are formed and transport these receptor proteins directly to the lysosomes for degradation.
- Organelles such as ribosomes and protein components are cellular structures that are sent to the lysosomes via the formation of autophagic vacuoles that are packed with cytoplasmic entities to be degraded.
- Endocytosed components that aren’t intended for recycling through the trans-Golgi processes are also transported to lysosomes through late endosomes.
Clinical correlation
Defective lysosomes can be noticed in different diseases including Rheumatoid arthritis, Gout, Silicosis, Asbestosis, Black lung disease, Byssinosis (cotton dust), Acidosis, Anoxia, Chronic granulomatous, etc.
References
- The Cell: A Molecular Approach. 2nd edition.
- Andrea Ballabio. The awesome lysosome. EMBO Mol Med. 2016 Feb; 8(2): 73–76.
- Haoxing Xu and Dejian Ren. Lysosomal Physiology. Annu Rev Physiol. 2015; 77: 57–80.
- Lysosomal membrane dynamics: structure and interorganellar movement of a major lysosomal membrane glycoprotein. J Cell Biol. 1986 May 1; 102(5): 1593–1605.
- Alberts B, Johnson A, Lewis J, et al. Transport from the Trans Golgi Network to Lysosomes. Molecular Biology of the Cell. 4th edition. New York: Garland Science; 2002.
- Frances M. Platt, Barry Boland, Aarnoud C. van der Spoel. Lysosomal storage disorders: The cellular impact of lysosomal dysfunction. J Cell Biol. 2012 Nov 26; 199(5): 723–734.
- Debjyoti Bandyopadhyay, Austin Cyphersmith, Jairo A. Zapata, Y. Joseph Kim, and Christine K. Payne. Lysosome Transport as a Function of Lysosome Diameter. PLoS One. 2014; 9(1): e86847.
- Paul Luzio, Yvonne Hackmann, Nele M.G. Dieckmann, and Gillian M. Griffiths. The Biogenesis of Lysosomes and Lysosome-Related Organelles. Cold Spring Harb Perspect Biol. 2014 Sep; 6(9): a016840.
- J.G. Gindhart, K.P. Weber, 2009. Lysosome and Endosome Organization and Transport in Neurons. Encyclopedia of Neuroscience.
Read also