Metformin is an anti-diabetic drug that has been taken by millions to control the effects of metabolic syndrome, e.g., diabetes. It is mainly used for reducing blood sugar levels in diabetic patients, but it is also used for other activities.
Physiologically, metformin has been shown to reduce glucose production in the liver.
Metformin increases insulin sensitivity, decreases intestinal glucose absorption, increases fatty acid oxidation, and reduces appetite, which all lead to weight loss. Thus, metformin has been shown to be the regulator of glucose-mediated effects in diabetic patients.
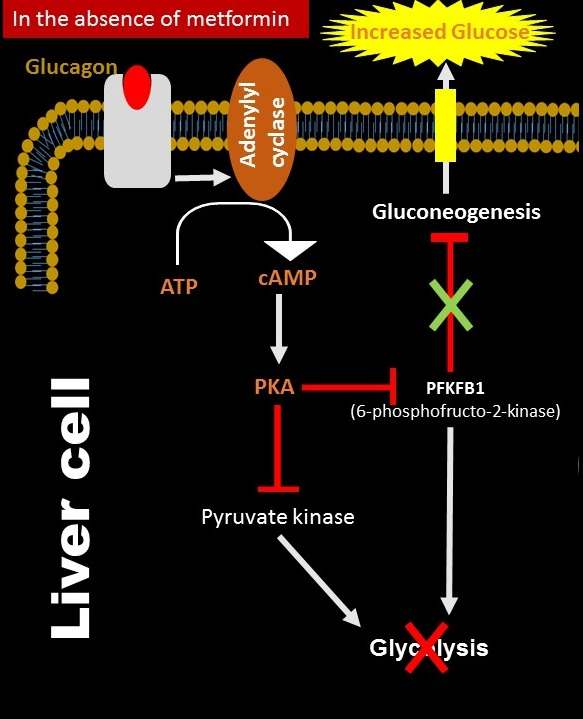
Before going to the metformin mechanism of action, first, we see the fate of a liver cell in the absence of metformin.
In general, glucagon binds its receptor and activates the enzyme, adenylate cyclase. The activated enzyme produces a second messenger, cAMP, using cellular ATP. The produced cAMP activates protein kinase A (PKA). The activated PKA binds and inhibits two enzymes, such as 6-Phosphofructo-2-Kinase (PFKFB1) and pyruvate kinase, which are responsible for the glycolysis process. Inactive forms of these enzymes fail to carry out glycolysis (glucose-lowering process), while, inactive forms of PFKFB1 are unable to control gluconeogenesis, which results in increased blood glucose levels.
Now we see the activities of a liver cell in the presence of metformin.
In general, uptake of metformin into liver cells is mediated by an organic cation transporter-1 (OCT1).
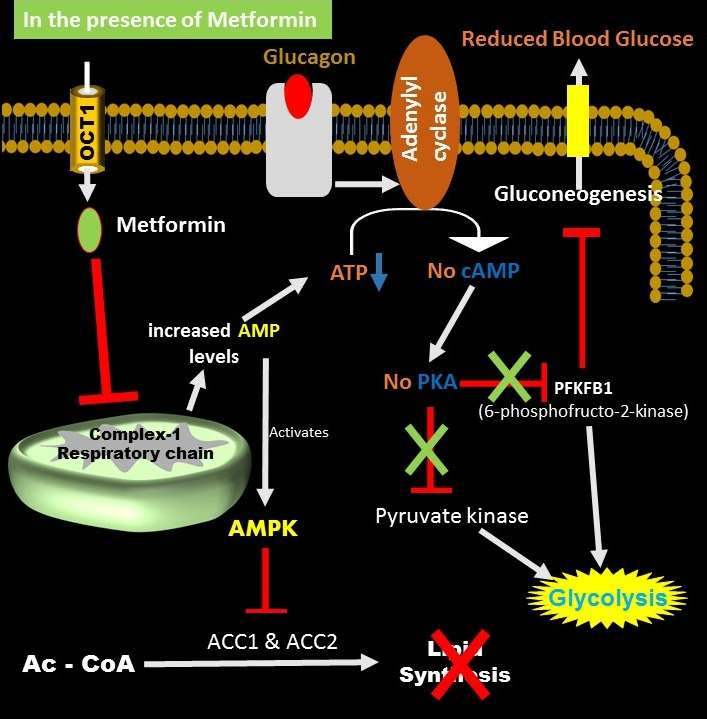
Once the metformin enters the cell, it reaches the mitochondrial matrix. In mitochondria, metformin inhibits complex 1 of the mitochondrial respiratory chain, resulting in a reduction in the production of the cellular energy molecule, ATP.
Even though, the glucagon binds its receptor and activates adenylate cyclase, the lower levels of ATP and higher levels of ADP and AMP in the liver cell reduce the activity of adenylate cyclase, which leads to a reduction in the production of the second messenger, cAMP.
The lower levels of cAMP can no longer activate PKA in the liver cells.
The inactive form of PKA is unable to inhibit the enzymes, PFKFB1 and Pyruvate kinase, resulting in a higher degree of glycolysis process. While the free form of PFKFB1 reduces gluconeogenesis, thereby reducing blood glucose levels.
Moreover, higher levels of cAMP can phosphorylate and activate an enzyme, AMP-activated protein kinase (AMPK).
The activated AMPK phosphorylates ACC1 and ACC2, which are two isoforms of the enzyme, acetyl-CoA carboxylase (ACC) involved in lipid synthesis and converts acetyl-CoA to malonyl-CoA (an intermediate molecule of lipid biosynthesis).
The phosphorylated ACC1 and ACC2 are no longer involved in lipid biosynthesis. Moreover, AMPK also promotes fat oxidation and reduces hepatic lipid storage, which results in the enchantment of hepatic insulin sensitivity.
Conclusion
Overall, the administration of metformin in diabetic patients lowers gluconeogenesis, reduces lipid biosynthesis, increases glycolysis, and promotes weight loss.
References:
Mariël F. van Stee, Albert A. de Graaf & Albert K. Groen. Actions of metformin and statins on lipid and glucose metabolism and possible benefit of combination therapy. Cardiovascular Diabetology.
Lilian Beatriz Aguayo Rojas & Marilia Brito Gomes. Metformin: an old but still the best treatment for type 2 diabetes. Diabetology & Metabolic Syndrome.
Yi-Wei Wang, Si-Jia He, Xiao Feng, Jin Cheng, Yun-Tao Luo, Ling Tian, and Qian Huang. Metformin: a review of its potential indications. Drug Des Devel Ther.
Graham Rena, D. Grahame Hardie, and Ewan R. Pearson. The mechanisms of action of metformin. Diabetologia.